Ethics Review Procedures in UK Healthcare PhD Dissertations: Compliance and Challenges
Ethics Review Procedures in UK Healthcare PhD Dissertations: Compliance and Challenges
- Home
- How To Article
- Ethics Review Procedures in UK Healthcare PhD Dissertations: Compliance and Challenges
Table of Content
- Ethical Governance in UK Healthcare Research
- Stages of the Ethics Review Process
- Key Ethical Requirements in Healthcare PhD Research
- Common Compliance Challenges Faced by PhD Students
- Ethical Risk Assessment in Healthcare Research
- Documentation Required for Ethics Approval
- Strategies for Ensuring Ethical Compliance
- Conclusion

Ethics Review Procedures in UK Healthcare PhD Dissertations: Compliance and Challenges
Healthcare research carried out in the United Kingdom is subject to a set of rigid ethical and regulatory guidelines. These guidelines are intended to ensure the protection of research participants, scientific integrity, and compliance with legislative requirements. For any PhD student intending to undertake clinical research, medical research, or other forms of phd research, it is a requirement to obtain Ethics Approval in UK PhD Healthcare Dissertation before commencing any research process. Ethics review procedures for healthcare research are very rigorous, especially when research involves vulnerable populations. [1]
Despite the availability of formal guidelines on ethics review procedures, many doctoral students often encounter difficulties in understanding these procedures and their compliance requirements. Understanding these procedures is essential for ensuring compliance with ethical guidelines for clinical research, medical research, and healthcare-related phd research conducted within UK doctoral programs such as phd in clinical psychology, phd in healthcare management, or related fields connected to a master’s degree in public health.[2]
1. Ethical Governance in UK Healthcare Research
Ethical governance of healthcare research in the UK is mainly governed by national regulatory bodies and university ethics committees. Healthcare phd research often requires approval from multiple regulatory authorities, depending on the nature of the clinical research or medical research involved.
- NHS Research Ethics Committees (REC)
- Health Research Authority (HRA)
- University Institutional Review Boards (IRB) or Ethics Committees
- General Data Protection Regulation (GDPR)
- Good Clinical Practice (GCP) Guidelines.
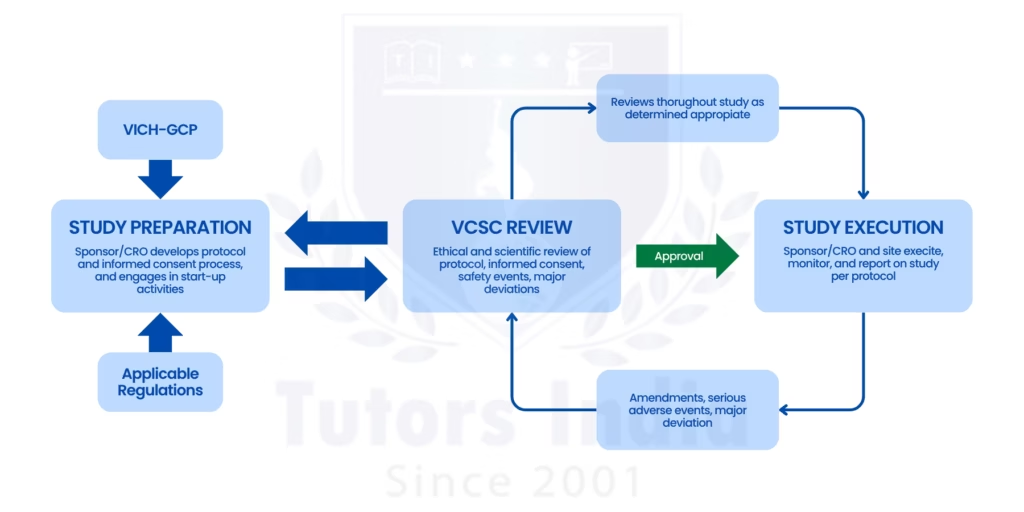
2. Stages of the Ethics Review Process
The ethics review process for healthcare PhD dissertations is often a structured process, and this process is aimed at evaluating the risk involved in the research.
- Development of research protocol
- Risk assessment in relation to the participants
- Preparation of ethics application documents
- Submission to university or the NHS ethics committee
- Review and feedback from the ethics panel
- Approval, modification requests, or rejection
The review process may take several weeks or months depending on the complexity of the clinical research or medical research involved.[3]
3. Key Ethical Requirements in Healthcare PhD Research
Healthcare research must follow very stringent ethical principles to ensure the protection of participants as well as the research itself. [4]
- Obtaining informed consent from all participants
- Maintaining confidentiality of patient information
- Minimising risks in clinical or behavioural studies
- Proper management and storage of research data
- Being transparent in research goals and methods
These ethical requirements are particularly important for research conducted within doctoral programs such as phd in clinical psychology, phd in healthcare management, and advanced public health studies following a master’s degree in public health.
4. Common Compliance Challenges Faced by PhD Students
Despite the presence of clear ethical guidelines, doctoral students often encounter several compliance challenges while conducting medical research or clinical research.
- Understanding complex ethical guidelines
- Developing participant information sheets
- Developing data management plans under GDPR
- Delays in the process due to incomplete ethical applications
- Working with multiple bodies, including the NHS and the university
In healthcare research, there is often the need to work with several review bodies, including institutional and national review bodies.[5]
5. Ethical Risk Assessment in Healthcare Research
Ethics committees evaluate healthcare clinical research and medical research based on defined risk categories to determine whether participants are adequately protected.[6]
| Risk Category | Description | Example |
| Low Risk | Minimal impact on participants | Anonymous health surveys |
| Moderate Risk | Potential psychological or behavioural effects | Interviews about patient experiences |
| High Risk | Clinical or intervention-based research | Drug trials or medical testing |
Researchers must clearly justify how potential risks will be mitigated before approval is granted.
6. Documentation Required for Ethics Approval
There are several supporting documents required by healthcare PhD students while applying for ethical approval. Some of these are:
- Research protocol
- Participant information sheet
- Informed consent form
- Data management plan
- Risk assessment documentation
- Recruitment strategy
Inadequate preparation of these documents is one of the major causes of delays/rejection of ethical approval applications.
7. Strategies for Ensuring Ethical Compliance
To successfully go through the ethics review processes, it is recommended that PhD researchers take a proactive approach to ethical compliance. The recommended strategies for this include: [7]
- Seeking the advice of the supervisor at the early stage of the research design
- Studying the university and NHS ethics guidelines
- Attending research ethics workshops
- Completing clear and comprehensive ethics applications
- Carrying out pilot studies where necessary.
These practices are particularly important for students pursuing advanced healthcare degrees such as phd in clinical psychology, phd in healthcare management, or research following a master’s degree in public health.
Conclusion
Ethics review procedures form an integral part of healthcare-based phd research in the UK. These procedures ensure that clinical research and medical research involving human participants meet ethical, legal, and scientific standards.
Understanding the governance structures, preparing detailed ethics applications, and engaging early with supervisors and ethics committees can help doctoral students successfully obtain Ethics Approval in UK PhD Healthcare Dissertation. By following these procedures carefully, researchers working in fields such as phd in clinical psychology, phd in healthcare management, and master’s degree in public health can ensure ethical compliance and contribute valuable research to the healthcare sector.
Ethics Review Procedures in UK Healthcare PhD Dissertations: Compliance and Challenges [Talk to a Dissertation Expert | Book a Free 15-Minute Consultation]
References
- Salifu Y. (2025). Ethical challenges in sensitive research: a reflective narrative on managing the clinician-researcher dual role. BMC palliative care, 24(1), 205. https://doi.org/10.1186/s12904-025-01850-y
- van Teijlingen, E. R., Douglas, F., & Torrance, N. (2008). Clinical governance and research ethics as barriers to UK low-risk population-based health research? BMC public health, 8, 396. https://doi.org/10.1186/1471-2458-8-396
- Capili, B., & Anastasi, J. K. (2024). Ethical Research and the Institutional Review Board: An Introduction. The American journal of nursing, 124(3), 50–54. https://doi.org/10.1097/01.NAJ.00010
- Das, N. K., & Sil, A. (2017). Evolution of Ethics in Clinical Research and Ethics Committee. Indian journal of dermatology, 62(4), 373–379. https://doi.org/10.4103/ijd.IJD_271
- Robishaw, J. D., DeMets, D. L., Wood, S. K., Boiselle, P. M., & Hennekens, C. H. (2020). Establishing and Maintaining Research Integrity at Academic Institutions: Challenges and Opportunities. The American journal of medicine, 133(3), e87–e90. https://doi.org/10.1016/j.amjmed.2019
- Patrick Hill T. (2014). Risk assessment in clinical trials: it don’t mean an ethical thing if it ain’t got that probability ring!. Ecancermedicalscience, 8, ed42. https://doi.org/10.3332/ecancer.2014
- McIntosh, T., Sanders, C., & Antes, A. L. (2020). Leading the people and leading the work: Practical considerations for ethical research. Translational issues in psychological science, 6(3), 257–270. https://doi.org/10.1037/tps0000260
